Lanthanide-induced conformational change of methanol dehydrogenase involving coordination change of cofactor pyrroloquinoline quinone†
Abstract
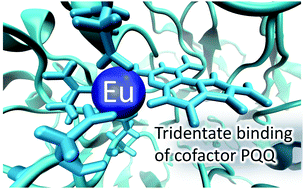
There is emerging interest in the role of lanthanides as cofactors for XoxF-type methanol dehydrogenase (MDH). Here, classical molecular dynamics simulations combined with fragment molecular orbital calculations were employed to rationalize the enzymatic activities of MDH (both XoxF- and MxaF-types) carrying different lanthanides. In XoxF-type MDH, lanthanide binding to cofactor pyrroloquinoline quinone was found to switch from tridentate to unidentate fashion as it switches from lighter to heavier lanthanides. This fact possibly plays a crucial role in the enzymatic activity exclusive to XoxF-type MDH incorporating lighter lanthanides.


 Please wait while we load your content...
Please wait while we load your content...