Tailored metastable Ce–Zr oxides with highly distorted lattice oxygen for accelerating redox cycles†
Abstract
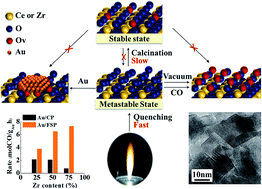
Ceria-based catalysts are widely used in oxidation or oxidation–reduction reactions in the field of environmental science. Their catalytic functions are determined by their ability to exchange oxygen species with oxidants. The enhancement of oxygen release is desired since it is often the rate-determining step in redox cycles. Herein, we developed a lattice oxygen distortion method to enhance oxygen activation by quenching the Ce–Zr oxide nanoparticles formed from an extremely high temperature. This process can ensure the formation of solid solutions as well as avoiding atomic rearrangement during calcination, retaining the lattice oxygen at a metastable and disordered state without vacancies. Reduction, vacuum or metal deposition will easily induce oxygen release accompanied by vacancy creation. The metastable oxides can provide about 19 times more oxygen vacancies than traditional ones in a CO atmosphere. CO oxidation rates increased with increasing Zr content from 25 to 75% and achieved a new level, which is attributed to the acceleration of oxygen circulation via promoting oxygen release and supplying plenty of oxygen vacancies for redox cycles. This strategy is expected to be applied in the design and fabrication of improved oxygen storage materials.


 Please wait while we load your content...
Please wait while we load your content...