Room temperature olefination of methane with titanium–carbon multiple bonds†
Abstract
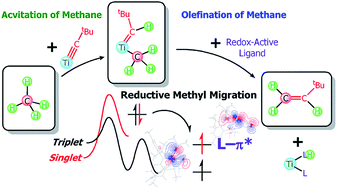
C–H activation of methane followed by dehydrocoupling at room temperature led ultimately to the formation of the olefin H2C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHtBu via the addition of redox-active ligands (L) such as thioxanthone or 2,2′-bipyridine (bipy) to (PNP)Ti
CHtBu via the addition of redox-active ligands (L) such as thioxanthone or 2,2′-bipyridine (bipy) to (PNP)Ti![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHtBu(CH3) (1). Using both of these exogenous ligand systems, we could trap the titanium fragment via an insertion reaction with these two substrates to afford species of the type (PNP)Ti(L)(LH). A combination of computational and isotopic labeling studies reveals that the L ligand promotes the C–C bond forming step by migration of the methyl moiety in 1 to the α-alkylidene carbon by producing a Ti(III) species (PNP)Ti{CH(CH3)tBu}(L). In the case of L = thioxanthone, β-hydrogen abstraction gives an olefin, whereas with 2,2′-bipyridine β-hydride elimination and migratory insertion lead to (PNP)Ti(L)(LH). These redox-active ligands play two important roles: (i) they accept an electron from the Ti-alkylidene fragment to allow the methyl to approach the alkylidene and (ii) they serve as traps of a hydrogen atom resulting from olefin elimination. These systems represent the first homogeneous models that can activate methane and selectively dehydrocouple it with a carbene to produce an olefin at room temperature.
CHtBu(CH3) (1). Using both of these exogenous ligand systems, we could trap the titanium fragment via an insertion reaction with these two substrates to afford species of the type (PNP)Ti(L)(LH). A combination of computational and isotopic labeling studies reveals that the L ligand promotes the C–C bond forming step by migration of the methyl moiety in 1 to the α-alkylidene carbon by producing a Ti(III) species (PNP)Ti{CH(CH3)tBu}(L). In the case of L = thioxanthone, β-hydrogen abstraction gives an olefin, whereas with 2,2′-bipyridine β-hydride elimination and migratory insertion lead to (PNP)Ti(L)(LH). These redox-active ligands play two important roles: (i) they accept an electron from the Ti-alkylidene fragment to allow the methyl to approach the alkylidene and (ii) they serve as traps of a hydrogen atom resulting from olefin elimination. These systems represent the first homogeneous models that can activate methane and selectively dehydrocouple it with a carbene to produce an olefin at room temperature.


 Please wait while we load your content...
Please wait while we load your content...