A vanillin-based copper(ii) metal complex with a DNA-mediated apoptotic activity†
Abstract
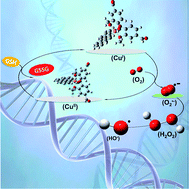
Vanillin (vanH) is the major component of vanilla and one of the most widely used flavoring agents. In this work the complex [Cu(phen)(van)2] was prepared and characterized by structural (X-ray), spectroscopic (IR, UV-Vis, EPR) and electrochemical techniques. This compound showed an octahedral geometry with an unusual arrangement of the vanillin ligands, where the methoxy groups of the vanillinate ions are coordinated opposite to each other. The compound promoted DNA cleavage in the presence of glutathione (GSH) and H2O2. At 40 μmol L−1 of complex with GSH (10 mmol L−1), there is a complete cleavage of DNA to nicked form II, while only at 10 μmol L−1 of this complex with H2O2 (1 mmol L−1) an extensive cleavage leading to form III took place. Additionally, we have evidences of superoxide generation upon reaction with GSH. Therefore, DNA fragmentation occurs likely through an oxidative pathway. MTT assays indicated that the complex is highly cytotoxic against three distinct cell lines: B16–F10 (IC50 = 3.39 ± 0.61 μmol L−1), HUH-7 (IC50 = 4.22 ± 0.31 μmol L−1) and 786-0 (IC50 = 10.38 ± 0.91 μmol L−1). Flow cytometry studies conducted with 786-0 cell line indicated cell death might occur by apoptosis. Cell cycle progression evaluated at 5 and 10 μmol L−1 resulted in a clear increase of 786-0 cells at G1 phase and depletion of G2/M, while higher doses showed an expressive increase of sub-G1 phase. Altogether, these results pointed out to a promising biological activity and potential as an anti-cancer agent.


 Please wait while we load your content...
Please wait while we load your content...