Deconvoluting the dual hypoglycemic effect of wedelolactone isolated from Wedelia calendulacea: investigation via experimental validation and molecular docking†
Abstract
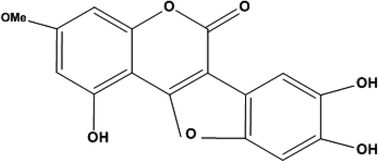
Wedelia calendulacea has a long history of use in the Indian Ayurvedic System of Medicine for the treatment, prevention, and cure of a diverse range of human diseases such as diabetes obesity, and other metabolic diseases. A wide range of chemical constituents, such as triterpenoid saponin, kauren diterpene, and coumestans, has been isolated from the plant. Conversely, no published literature is available in relation to the isolation of wedelolactone (WEL) for its anti-diabetic effect. The aim of the present study was to isolate the bioactive phyto-constituent from Wedelia calendulacea and to scrutinize the antidiabetic effect with its possible mechanism of action. The structure of the isolated compound was elucidated by different spectroscopy techniques. Proteins, such as dipeptidyl peptidase-4 (DPPIV), glucose transporter 1 (GLUT1), and peroxisome proliferator-activated receptors-γ (PPARγ), were also subjected to in silico docking. Later, this isolated compound was scrutinized against α-glucosidase and α-amylase enzyme activity along with an oral glucose tolerance test (OGTT) for estimation of glucose utilization. Streptozotocin (STZ) was used for the induction of type II diabetes mellitus (DM) in Wistar rats. The rats were divided into different groups and received the WEL (5, 10, and 20 mg kg−1, b.w.) and glibenclamide (2.5 mg kg−1, b.w.) for 28 days. The blood glucose level (BGL), plasma insulin, and body weight were determined at regular time intervals. The serum lipid profile hypolipidemic effect for the different antioxidant markers and hepatic tissue markers were scrutinized along with an inflammatory mediator to deduce the possible mechanism. With the help of spectroscopy techniques, the isolated compound was identified as wedelolactone. In the docking study, WEL showed docking scores of −6.17, −9.43, and −7.66 against DPP4, GLUTI, and PRARY, respectively. WEL showed the inhibition of α-glucosidase (80.65%) and α-amylase (93.83%) and suggested an effect on postprandial hyperglycemia. In the OGTT, WEL significantly (P < 0.001) downregulated the BGL, a marker for better utilization of drugs. In the diabetes model, WEL reduced the BGL and enhanced the plasma insulin and body weight. It also significantly (P < 0.001) modulated the lipid profile; this suggested an anti-hyperlipidemia effect. WEL significantly (P < 0.001) distorted the hepatic tissue, acting as an antioxidant marker in a dose-dependent manner. WEL significantly (P < 0.001) downregulated the C-reactive protein (CRP), tumor necrosis factor alpha (TNF-α), and interleukin 6 (IL-6) level. On the basis of the available results, we can conclude that WEL can be an alternative drug for the treatment of type II DM either by inhibiting the production of inflammatory mediator or by the downregulation of oxidative stress.


 Please wait while we load your content...
Please wait while we load your content...