The fabrication of amphiphilic double dynamers for responsive Pickering emulsifiers†
Abstract
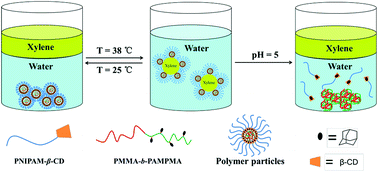
Amphiphilic graft copolymers exhibiting both supramolecular and molecular dynamicities and their application as responsive Pickering emulsifiers were demonstrated in the present study. Amphiphilic dynamers were fabricated by the introduction of dynamic imine bonds into supramolecular polymers built on host–guest interactions between β-cyclodextrin (β-CD) and adamantane. For this purpose, well-defined diblock copolymers poly(methyl methacrylate)-b-poly(((4-adamantaneimino)methyl)phenyl methacrylate) (PMMA-b-PAMPMA) containing reversible C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N covalent linkages and β-CD terminated poly(N-isopropylacrylamide) (PNIPAM-β-CD) polymers were firstly synthesized by atom transfer radical polymerization (ATRP) and a combination of reversible addition–fragmentation chain transfer (RAFT) polymerization and click reaction, respectively. Hydrophilic PNIPAM polymers were then grafted onto PMMA-b-PAMPMA backbone by means of the host–guest inclusion interaction between the adamantane lateral groups of backbone precursors and the β-CD end groups of PNIPAM-β-CD side-chain precursors in THF, yielding amphiphilic PMMA-b-PAMPMA-g-PNIPAM graft copolymers with both supramolecular and molecular dynamicities. The amphiphilic double dynamers further self-assembled to polymeric aggregates in the mixture of THF/water, and nearly monodispersed spherical core–shell particles were achieved after the removal of THF by dialysis. The hydrophobic PMMA-b-PAMPMA and the hydrophilic PNIPAM-β-CD acted as the cores and the shells, respectively. These self-assembled polymer particles exhibited morphological transitions in response to external stimuli, such as temperature and pH. They also displayed significant emulsifying performance and stimuli-responsive interfacial activities, making them good candidates for responsive Pickering emulsifiers.
N covalent linkages and β-CD terminated poly(N-isopropylacrylamide) (PNIPAM-β-CD) polymers were firstly synthesized by atom transfer radical polymerization (ATRP) and a combination of reversible addition–fragmentation chain transfer (RAFT) polymerization and click reaction, respectively. Hydrophilic PNIPAM polymers were then grafted onto PMMA-b-PAMPMA backbone by means of the host–guest inclusion interaction between the adamantane lateral groups of backbone precursors and the β-CD end groups of PNIPAM-β-CD side-chain precursors in THF, yielding amphiphilic PMMA-b-PAMPMA-g-PNIPAM graft copolymers with both supramolecular and molecular dynamicities. The amphiphilic double dynamers further self-assembled to polymeric aggregates in the mixture of THF/water, and nearly monodispersed spherical core–shell particles were achieved after the removal of THF by dialysis. The hydrophobic PMMA-b-PAMPMA and the hydrophilic PNIPAM-β-CD acted as the cores and the shells, respectively. These self-assembled polymer particles exhibited morphological transitions in response to external stimuli, such as temperature and pH. They also displayed significant emulsifying performance and stimuli-responsive interfacial activities, making them good candidates for responsive Pickering emulsifiers.


 Please wait while we load your content...
Please wait while we load your content...