Boron-doped graphene as a promising electrocatalyst for NO electrochemical reduction: a computational study†
Abstract
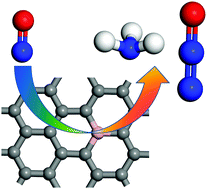
The NO electrochemical reduction (NOER) holds great promise for the conversion of harmful NO species in groundwater to useful fuels and chemicals. However, it is still challenging to achieve the large-scale practical application of NOER if high-priced Pt-based electrocatalysts cannot be replaced with other high efficient, low cost, and stable catalysts. Here, by means of density functional theory (DFT) computations, the potential of boron-doped graphene (BG) as a metal-free electrocatalyst for the NOER was systemically explored. Our results revealed that the introduction of a B atom into graphene enhances its interaction with the HNO* intermediate, effectively promoting the subsequent NOER steps to proceed though the NH2O*, NH2OH* and NH2* intermediates with a low limiting potential of −0.35 V, and NH3 is the main product. Furthermore, the HNO* intermediate provides active sites for coupling with NO to generate N2O. Therefore, B-doped graphene is a quite promising metal-free electrocatalyst with high-efficiency for NO reduction and conversion.


 Please wait while we load your content...
Please wait while we load your content...