Optimization of the selectivity and rate of copper radioisotope complexation: formation and dissociation kinetic studies of 1,4,8-trimethylcyclam-based ligands with different coordinating pendant arms†
Abstract
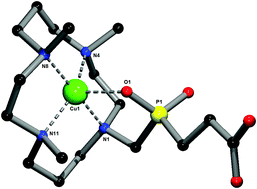
Selectivity and rate of complex formation with metal radionuclides are crucial parameters for the utilization of ligating systems in nuclear medicine. One of the very suitable metals used in these applications is copper, which has a number of radioisotopes with useful properties. The thermodynamic and kinetic properties of Cu(II) complexes with 10 macrocyclic 1,4,8-trimethylcyclam-based ligands having one coordinating acid pendant arm (Me3cyclam–R) were investigated in solution, allowing a direct comparison of the influence of the particular pendant arm on the properties of the complexes. They include the derivative with R = CH2CO2H (HL1) and a family containing various phosphorus acids R = CH2P(O)(OH)–X, where X = OEt (HL2); X = OH (H2L3); X = H (HL4); X = CH2CH2CO2H (H2L5); X = CH2P(O)(H)OH (H2L6); X = CH2P(O)(OH)2 (H3L7); X = CH2N(CH2C6H5)2 (HL8); X = CH2NH2 (HL9) and X = CH2N(CH2CO2H)2 (H3L10). For comparison, 1,4,8,11-tetramethylcyclam (TMC: R = Me) was used. The formation kinetics showed that ligands endowed with a coordinating pendant arm bind Cu(II) ions much faster when compared to TMC. At pH < 4, the fastest complexation was observed for acetate derivative HL1. At higher pH and, especially, at pH relevant for living and biocompatible systems (pH ≈ 6–7), the ligands with methylene(phosphonatomethyl)phosphinate and methylenephosphonate pendant arms (H3L7 and H2L3) showed the fastest complexation. Acid-assisted dissociation of Cu(II) complexes with the ligands endowed with a coordinating pendant arm is similar for all studied systems (τ1/2 = 7–35 min, 1 M HClO4, 25 °C). In contrast, the inertness of the Cu(II)–TMC complex is much lower (τ1/2 = 22 s under the same conditions). Potentiometric study of the selected ligands has confirmed a high thermodynamic selectivity of the studied ligands for Cu(II) binding over complexation of Ni(II) and Zn(II) (the differences between the stability constants reach 6–7 orders of magnitude). Fast complexation of Cu(II) at radio-level concentrations was observed, showing that the best ligands for potential in vivo use are those containing phosphonate or mixed geminal phosphinate–phosphonate pendant groups. One of the ligands (H2L3) and three Cu(II) complexes (of H2L5, H3L7 and H3L10) were structurally characterized by means of X-ray diffraction study. The predicted conformation I of the macrocycle was confirmed in all three complexes.


 Please wait while we load your content...
Please wait while we load your content...