Unlocking the photobiological conversion of CO2 to (R)-3-hydroxybutyrate in cyanobacteria†
Abstract
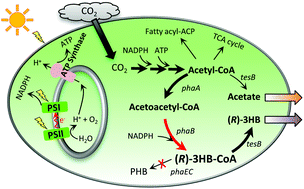
Escalating concerns about CO2 emission from fossil fuel utilization and environmental pollution from fossil-derived plastic waste call for the sustainable production and utilization of renewable biodegradable plastic materials. Polyhydroxyalkanoates (PHAs) are biodegradable and biocompatible thermoplastics with thermal and mechanical properties comparable to conventional plastics; thus, they are promising materials to mitigate environmental pollution. (R)-3-Hydroxybutyrate (3HB), which is the most common building-block for PHAs, has the potential to be utilized in various medical applications and can also serve as a precursor to a variety of value-added stereospecific chemicals. In addition, it can be produced by microorganisms, such as engineered cyanobacteria, from inexpensive renewable resources such as waste CO2. However, higher titer and rate of (R)-3HB production by cyanobacteria beyond that found in the current literature are critical for commercial applications. Herein, we employed a facile strategy to identify the rate-limiting step in the photoautotrophic production of (R)-3HB by the cyanobacterium Synechocystis and found that acetoacetyl-CoA reductase activity is the bottleneck in the process. Optimization of the gene's ribosome binding site led to a 2.2-fold increase in enzyme activity. In the culture medium of the engineered organism, the (R)-3HB titer reached 1.84 g L−1 within 10 days, with a peak productivity of 263 mg L−1 day−1, using CO2 and light as the sole carbon and energy sources. Moreover, dramatic changes in carbon partition were discovered in the (R)-3HB-producing cells along the course of cultivation using 13C-metabolic flux analysis; after the rapid growth phase, a majority of carbon flux was redirected from the cell mass formation to the production of (R)-3HB in the engineered Synechocystis under the examined experimental conditions.


 Please wait while we load your content...
Please wait while we load your content...