Structure and dynamics of water on the forsterite surface†
Abstract
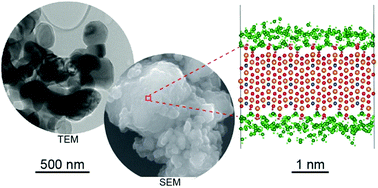
The behavior of water on mineral surfaces is the key to understanding interfacial and chemical reaction processes. Olivine is one of the major rock-forming minerals and its interaction with water is a ubiquitous phenomenon both on Earth's surface and in the subsurface. This work presents a combined study using molecular dynamics (MD) simulations and quasi-elastic neutron scattering (QENS) experiments conducted using three different instruments to study the structure and dynamics of water on the forsterite (Mg-end member of olivine) surface at 270 K. A combination of three different QENS instruments probes dynamical processes occurring across a broad range of time scales (∼1 ps to ∼1 ns in this study). The water structure on the hydroxylated surface is composed of three distinct water layers, transitioning from well-ordered and nearly immobile closest to the surface to a less structured layer. The energies of three motions (including translation and rotation) derived from simulations agree well with the experiments, covering the energy range from a few to hundreds of micro electron volts.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...