Factors governing when a metal-bound water is deprotonated in proteins†
Abstract
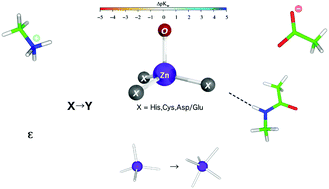
Understanding when a metal-bound water molecule in a protein is deprotonated is important as this affects the charge distribution in the metal-binding/enzyme active site and thus their interactions, the enzyme mechanism, and inhibitor design. The protonation state of the metal-bound water molecule at a given pH depends on its pKa value, which in turn depends on the properties of the cation, its ligands, and the protein environment. Here, we reveal how and the extent to which (i) the first-shell composition (type, charge, and number of ligands), (ii) the metal site's immediate surroundings (first-shell⋯second-shell hydrogen-bonding interactions, metal–ligand distance constraints, and ligand-binding mode) and (iii) the protein architecture and coupled solvent interactions (long-range electrostatic interactions and solvent exposure of the site) affect the Zn2+-bound water pKa. The results, which are consistent with available experimental pKa values of Zn2+-bound water, provide guidelines to predict when Zn2+-bound water would likely be deprotonated at physiological pH.


 Please wait while we load your content...
Please wait while we load your content...