Incorporation of graphene nanodots and oxygen defects triggers robust coupling between solar energy and reactive oxygen†
Abstract
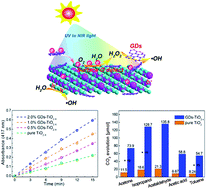
Efficient coupling between solar energy and active oxygen-related radicals plays a very important role in promoting photocatalytic oxidation processes; however, the poor solar energy harvesting, serious carrier recombination and especially sluggish interfacial reaction kinetics always make this process very inefficient. Here we report for the first time that the synergy mechanism between surface oxygen defects and graphene nanodots over the TiO2 photocatalyst boosts the solar-to-chemical energy conversion and hence enables the ultra-efficient photocatalytic decomposition of volatile organic compounds (VOCs). As a proof of concept, the surface oxygen defect incorporation not only can effectively suppress surface charge recombination, but also can promote the chemisorption of oxygen and hence enables robust H2O2 evolution over the TiO2 photocatalyst through visible–near-infrared illumination. The graphene nanodots (GDs), which were originally synthesized via an electrochemical tailoring method and possess excellent peroxidase activity can substitute Fe3+ to trigger robust solid–gas interfacial Fenton-like reaction (the rigorous reaction environment of the classic Fenton reaction greatly limits its implementation under ordinary air conditions). In addition, the excellent photophysical properties of GDs also can act as a sensitizer to further optimize the light harvesting performance of the resultant hybrid from the UV to NIR region. As a surprising achievement, the robust interfacial Fenton-like reaction, broader optical window and efficient charge separation endow the hybrid catalyst with ultra-efficient energy coupling between photons, excitons and oxygen-related radicals, and hence enable excellent photocatalytic oxidation activity for the decomposition of various VOCs.


 Please wait while we load your content...
Please wait while we load your content...