Alkaline niobates ANbO3 (A = Li, Na, K) as heterogeneous catalysts for dipropyl sulfide oxidation†
Abstract
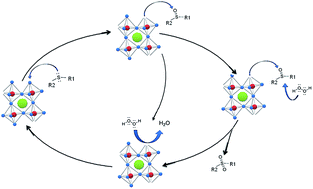
Alkaline ANbO3 (A = Li, Na, K) niobates with perovskite-type structure were prepared by a sol gel method, characterized by XRD, BET, TG/DTA, O2-TPD, DRS UV-Vis and FTIR and evaluated as heterogeneous catalysts for the selective oxidation of dipropyl sulfide (DPS) to its corresponding sulfoxide. The effects of different reaction parameters were evaluated, such as catalyst weight, the solvent and oxidant nature, reaction temperature and reuse. In order to gain better knowledge of the oxidation process, a kinetic study of the reaction was carried out. The global pseudo first order constant, the initial reaction rate and the apparent activation energy were calculated, and simplified reaction steps are proposed. KNbO3 showed the highest catalytic activity and sulfoxide selectivity with a periodic trend of KNbO3 > NaNbO3 ∼ LiNbO3.

 Please wait while we load your content...
Please wait while we load your content...