Hydrogels generated by low-molecular-weight PEGylated luteolin and α-cyclodextrin through self-assembly for 5-fluorouracil delivery†
Abstract
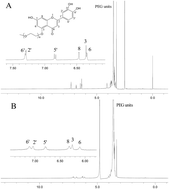
Hydrophobic luteolin (LUT) was conjugated to the oligomeric chain of methoxypoly(ethylene glycol) (mPEG) to form novel amphiphilic mPEG1900–LUT conjugates. Then mPEG1900–LUT, by using its adjacent 3′ and 4′ hydroxyl groups, were assembled with magnetic Fe3O4 particles in an aqueous solution to form mPEG1900–LUT–Fe3O4 conjugates, which formed hybrid Fe3O4 particles. The spectral properties and micellization of the conjugates were studied. Both mPEG1900–LUT and mPEG1900–LUT–Fe3O4 conjugates were able to self-assemble into stable supramolecular hydrogels through α-cyclodextrin (α-CD) in water, and their gelation times and temperatures were determined. The hydrogels displayed typical porous structures, which are suitable for drug delivery. Therefore, 5-fluorouracil (5-FU) was loaded into the formed hydrogels to control its release in vitro. The drug release observations showed that introducing Fe3O4 particles into the hydrogel improved the sustained release effect.

 Please wait while we load your content...
Please wait while we load your content...