Dipeptide-bonded stationary phases for hydrophilic interaction liquid chromatography†
Abstract
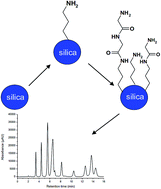
Dipeptides of glycine and alanine were directly synthesized onto a silica surface using an established procedure based on the fundamental principles of the Merrifield method. The peptide-silica stationary phases were prepared via a two-stage reaction including initial modification of the silica with γ-aminopropyltrimethoxysilane and subsequent bonding of the protected amino acids (alanine and glycine). The surface properties of the stationary phases, before and after chemical modification, were characterized by different physicochemical techniques. The resulting data provided information about the structure of the bonded ligands, the heterogeneity of the peptide layers, and a potential specification for the hydrophilic chromatography mechanism. Thereafter, the obtained packing materials were applied as stationary phases for the analysis of nucleic bases and nucleosides in HILIC mode. The application and comparison of the packing materials with a commercially available column was also performed. Both peptide-silica adsorbents were also compared in terms of their retention and separation selectivity of nucleosides and nucleic bases.

 Please wait while we load your content...
Please wait while we load your content...