Synthesis and hypergolic activity evaluation of some new ammonium-imidazolium based ionic liquids†
Abstract
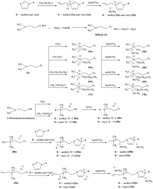
In this study, a new class of dicyanamide-based ionic liquids containing mono and dicationic counterparts was synthesized and characterized. Results demonstrate that the physical properties of these salts, including thermal stability, melting point, density and viscosity, are highly dependent on the structure of cation and type of anion. It was observed that the thermal stability of dicationic ionic liquids is higher for the monocationic ones. Furthermore, droplet tests were used to investigate the hypergolic properties of these ionic liquids. All of the synthesized compounds showed hypergolic activity upon contact with white fuming nitric acid, suggesting that some may have potential for application as bipropellants or energetic additives.

 Please wait while we load your content...
Please wait while we load your content...