Dramatically different charge transport properties of bisthienyl diketopyrrolopyrrole-bithiazole copolymers synthesized via two direct (hetero)arylation polymerization routes†
Abstract
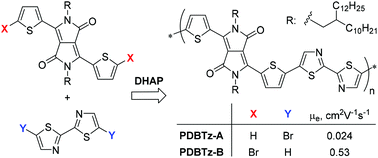
Two bisthienyl diketopyrrolopyrrole (DPP)-bithiazole based copolymers were synthesised via two different direct (hetero)arylation polymerization (DHAP) routes. When a bisthienyl DPP and 5,5′-dibromo-2,2′-bithiazole were used as monomers, the resulting polymer PA-1 showed poor solubility, ill-organized chain ordering and low performance in organic thin film transistors (OTFTs). Surprisingly, the synthetic route using a dibrominated bisthienyl DPP and 2,2′-bithiazole as monomers produced a polymer PB-1 with improved solubility, much higher crystallinity and excellent charge transport performance in OTFTs. The electron and hole mobilities of up to 0.53 cm2 V−1 s−1 and 0.06 cm2 V−1 s−1, respectively, achieved for PB-1 are more than one order of magnitude higher than those of PA-1 and also better than the mobilities reported for a similar polymer synthesized via Stille coupling polymerization. The dramatically different carrier mobilities observed for these two polymers are accounted for by their different amounts of α–β coupling linkages and branched (and lightly cross-linked) structure defects formed in the respective synthetic routes. This work also demonstrated for the first time that 2,2′-bithiazole is a suitable monomer for the construction of conjugated polymers with good electron transport performance via DHAP.

 Please wait while we load your content...
Please wait while we load your content...