Syntheses and catalytic activities of new metallodendritic catalysts
Abstract
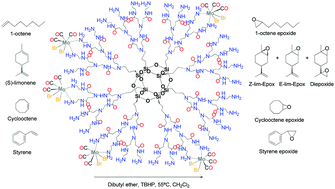
This investigation presents the syntheses of new metallodendritic catalysts from a silsesquioxane core (Silsesq-PrNH3+Cl−) via a new route. For optimization of the syntheses of the catalysts, reaction times, thermodynamic effects, and the amounts of [Mo(η3-C3H5)Br(CO)2(NCCH3)2] (1) and [Mo(CO)3Br2(NCCH3)2] (2) complexes fixed on the second-generation dendrimer (PDG2.0) were studied. After the optimization studies and with the objective of application in catalysis, the catalysts were prepared by reaction of PDG2.0 with the organometallic complexes and characterized by elemental analysis (CNH), FTIR, 13C and 29Si NMR, XRD, TGA, SEM, EDS and BET. The metallodendritic catalysts were tested in the epoxidation of olefins and shown to be a vigorous catalyst with conversion rates between 76 and 97% for PDG2.0–[(Mo(η3-C3H5)(CO)2Br)8] and 30–80% for PDG2.0–[(Mo(CO)3Br2)5], and a high selectivity in the formation of epoxides using tertbutyl hydroperoxide (TBHP) as an oxidant. The catalysts remain highly active after being used for at least 5 cycles. To the best of our knowledge, this study is the first to have reported the preparation of these new dendritic catalysts, as well as the catalytic comparison with their previous generations in the epoxidation of 1-octene, cyclooctene, (S)-limonene, cis-3-hexen-1-ol, trans-3-hexen-1-ol and styrene.

 Please wait while we load your content...
Please wait while we load your content...