Monitoring of chromosome dynamics of single yeast cells in a microfluidic platform with aperture cell traps†
Abstract
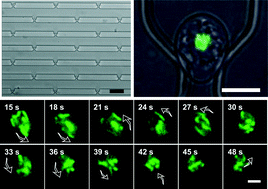
Chromosome movement plays important roles in DNA replication, repair, genetic recombination, and epigenetic phenomena during mitosis and meiosis. In particular, chromosome movement in the nuclear space is essential for the reorganization of the nucleus. However, conventional methods for analyzing the chromosome movements in vivo have been limited by technical constraints of cell trapping, cell cultivation, oxygenation, and in situ imaging. Here, we present a simple microfluidic platform with aperture-based cell trapping arrays to monitor the chromosome dynamics in single living cells for a desired period of time. Under the optimized conditions, our microfluidic platform shows a single-cell trapping efficiency of 57%. This microfluidic approach enables in situ imaging of intracellular dynamics in living cells responding to variable input stimuli under the well-controlled microenvironment. As a validation of this microfluidic platform, we investigate the fundamental features of the dynamic cellular response of the individual cells treated with different stimuli and drug. We prove the basis for dynamic chromosome movement in single yeast cells to be the telomere and nuclear envelope ensembles that attach to and move in concert with nuclear actin cables. Therefore, these results illustrate the monitoring of cellular functions and obtaining of dynamic information at a high spatiotemporal resolution through the integration of a simple microfluidic platform.

 Please wait while we load your content...
Please wait while we load your content...