Cationic mono and dicarbonyl pincer complexes of rhodium and iridium to assess the donor properties of PCcarbeneP ligands†
Abstract
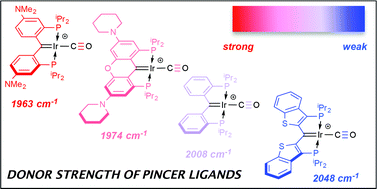
The donor properties of five different PCcarbeneP ligands are assessed by evaluation of the CO stretching frequencies in iridium(I) and rhodium(I) carbonyl cations. The ligands feature dialkyl phosphine units (R = iPr or tBu) linked to the central benzylic carbon by either an ortho-phenylene bridge, or a 2,3-benzo[b]thiophene linker; in the former, substituent patterns on the phenyl linker are varied. The carbonyl complexes are synthesized from the (PCcarbeneP)M–Cl starting materials via abstraction of the chlorides in the presence of CO gas. In addition to the expected mono carbonyl cations, products with two carbonyl ligands are produced, and for the rhodium example, a novel product in which the second carbonyl ligand adds reversibly across the Rh![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond to give an η2 ketene moiety was characterized. The IR data for the complexes shows the 2,3-benzo[b]thiophene linked system to be the poorest overall donor, while the phenyl bridged ligands incorporating electron donating dialkyl amino groups para to the anchoring carbene are very strongly donating pincer arrays.
C bond to give an η2 ketene moiety was characterized. The IR data for the complexes shows the 2,3-benzo[b]thiophene linked system to be the poorest overall donor, while the phenyl bridged ligands incorporating electron donating dialkyl amino groups para to the anchoring carbene are very strongly donating pincer arrays.

 Please wait while we load your content...
Please wait while we load your content...