Structural diversity of halocarbonyl molybdenum and tungsten PNP pincer complexes through ligand modifications†
Abstract
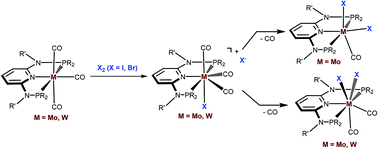
This work presents a comparative study of a series of halocarbonyl Mo(II) and W(II) complexes of the types [M(PNP)(CO)3X]X and [M(PNP)(CO)2X2] (M = Mo, W; X = I, Br), featuring PNP pincer ligands based on a 2,6-diaminopyridine scaffold. The complexes were prepared and fully characterized. The syntheses of these complexes were accomplished by treatment of [M(PNP)(CO)3] with stoichiometric amounts of I2 and Br2, respectively. The modification of the 2,6-diaminopyridine scaffold by introducing NMe and NPh instead of NH spacers with concomitant modification of the phosphine moieties changed the steric and electronic properties of the PNP ligand significantly. While in the case of NH linkers exclusively cationic seven-coordinate complexes of the type [M(PNP)(CO)3X]+ were obtained with NMe and NPh spacers neutral seven-coordinate complexes of the type [M(PNP)(CO)2X2] were afforded. In the case of the latter, when the reaction is performed in the presence of CO also [M(PNP)(CO)3X]+ complexes are formed which slowly lose CO to give [M(PNP)(CO)2X2]. The halocarbonyl tungsten chemistry parallels that of molybdenum. The only exception is molybdenum in conjunction with the PNPMe-iPr ligand, where the coordinatively unsaturated complex [Mo(PNPMe-iPr)(CO)X2] is formed. DFT mechanistic studies reveal that the seven-coordinate complexes should be the thermodynamic as well as the kinetic products. Since [Mo(PNPMe-iPr)(CO)X2] is the observed product it suggests that the reaction follows an alternative path. Structures of representative complexes were determined by X-ray single crystal analyses.

 Please wait while we load your content...
Please wait while we load your content...