Barium borohydride chlorides: synthesis, crystal structures and thermal properties†
Abstract
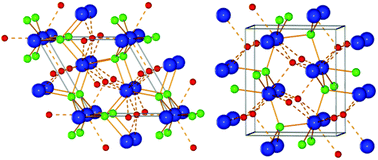
Here we report the synthesis, mechanism of formation, characterization and thermal decomposition of new barium borohydride chlorides prepared by mechanochemistry and thermal treatment of MBH4–BaCl2, M = Li, Na or K in ratios 1 : 1 and 1 : 2. Initially, orthorhombic barium chloride, o-BaCl2 transforms into o-Ba(BH4)xCl2−x, x ∼ 0.15. Excess LiBH4 leads to continued anion substitution and a phase transformation into hexagonal barium borohydride chloride h-Ba(BH4)xCl2−x, which accommodates higher amounts of borohydride, possibly x ∼ 0.85 and resembles h-BaCl2. Thus, two solid solutions are in equilibrium during mechano-chemical treatment of LiBH4–BaCl2 (1 : 1) whereas LiBH4–BaCl2 (2 : 1) converts to h-Ba(BH4)0.85Cl1.15. Upon thermal treatment at T > ∼200 °C, h-Ba(BH4)0.85Cl1.15 transforms into another orthorhombic barium borohydride chloride compound, o-Ba(BH4)0.85Cl1.15, which is structurally similar to o-BaBr2. The samples with M = Na and K have lower reactivity and form o-Ba(BH4)xCl2−x, x ∼ 0.1 and a solid solution of sodium chloride dissolved in solid sodium borohydride, Na(BH4)1−xClx, x = 0.07. The new compounds and reaction mechanisms are investigated by in situ synchrotron radiation powder X-ray diffraction (SR-PXD), Fourier transform infrared spectroscopy (FT-IR) and simultaneous thermogravimetric analysis (TGA), differential scanning calorimetry (DSC), mass spectroscopy (MS) and temperature programmed photographic analysis (TPPA).

 Please wait while we load your content...
Please wait while we load your content...