Transition metal promoted K/Mo2C as efficient catalysts for CO hydrogenation to higher alcohols
Abstract
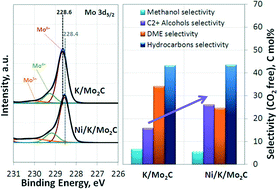
This study investigates the effect of transition metal promotion in a series of K/Mo2C catalysts doped with Ni, Cu and Mn for the hydrogenation of CO to higher alcohols. The catalysts were found to produce a significant amount of higher alcohols, consisting mainly of linear alcohols with two to four carbon atoms, at mild reaction conditions (temperature of 280 °C and pressure of 40–60 bar). All transition metals increased the selectivity for higher alcohols compared to the reference material. Nickel had the most favorable effect, as it greatly increased CO conversion as well. XPS results revealed a strong interaction between molybdenum in the carbidic phase and the transition metals, indicative of electron transfer from the dopants to the Mo atoms. In the Ni/K/Mo2C catalyst, the formation of a mixed Ni–Mo carbidic phase was evidenced. We postulate that this phase serves as the active centre for non-dissociative CO chemisorption and leads, in combination with dissociative CO adsorption on the Mo2C sites, to higher alcohol formation. Overall, the Ni/K/Mo2C catalyst exhibited the highest catalytic activity, with 23.2% CO conversion, 30.5% higher alcohol selectivity and space time yield to oxygenates of 147.4 mg gcatalyst−1 h−1 at 280 °C and 60 bar. Stability testing of the optimum catalyst for 400 hours time on stream demonstrated considerable deactivation, with an activity decrease of ~48%. The reduction in reactivity can be ascribed to the segregation of the mixed Ni–Mo carbidic phase and the formation of hydrated oxidic nickel species together with a well-defined K2Mo2O7 phase.

 Please wait while we load your content...
Please wait while we load your content...