Photoactivity and charge trapping sites in copper and vanadium doped anatase TiO2 nano-materials†
Abstract
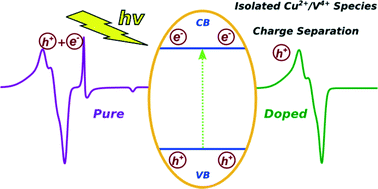
Two series of Cu2+ and V4+ doped anatase TiO2 samples were prepared using the microemulsion synthetic route by varying the metal/Ti ratio. The samples were characterized by UV-vis absorption, nitrogen physisorption, XRD, XPS, Raman and EPR spectroscopy. Their photocatalytic activity against toluene photo-oxidation was evaluated under both sunlight-type and pure UV light irradiation. The photoactivity was drastically affected by the dopant content. Low or moderate metal doping levels were beneficial for toluene photo-degradation, while high doping levels suppressed the photoactivity. Electron paramagnetic resonance (EPR) spectroscopy was employed to study the structure and electronic characteristics of the prepared catalysts and elucidate the physicochemical aspects governing the photoactivity. The presence of different Cu and V species was detected as a function of the metal content in the catalyst. EPR spectroscopy revealed that photoinitiated charge formation takes place in both the Cu and V doped TiO2 series and that charge separation may be enhanced depending on the dopant content. A direct correlation between the dopant species formed and hole formation was observed in both series. Photoactivity is directly correlated with the formation of holes, which in turn is tuned by the dopant content regulating the isolated metal centers versus metal cluster formation. Low or moderate doping levels enhanced the separation of photo-produced electron–hole pairs via accepting e−, eliminating trapping sites related to localized Ti3+ states and increasing the abundance of hole species. At higher levels where metal clusters prevailed, dopants acted as recombination centers, deteriorating the photoactivity in both TiO2-doped series. The results indicate a general mechanism potentially applicable to similar photocatalytic systems.

 Please wait while we load your content...
Please wait while we load your content...