Photoluminescence spectra and quantum yields of gold nanosphere monomers and dimers in aqueous suspension†
Abstract
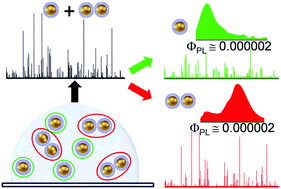
The intrinsic one-photon excited photoluminescence (PL) of dimers and monomers of gold spheres suspended in water was studied by combining photon time-of-flight spectroscopy (PTOFS) and light scattering fluctuation correlation spectroscopy (LS-FCS). The samples are obtained by precisely controlling the dimerization of aqueous colloidal systems based on 50 and 80 nm gold nanospheres. The combination of PTOFS and LS-FCS enables the separate spectroscopic study of monomers and dimers even though they exist as a mixture in the samples. PL emission spectra and diffusional dynamics are obtained simultaneously through measurement at the single particle level. The PL spectra resemble the light scattering spectra, indicating the plasmon-assisted character of the photoluminescence process. We determine the intrinsic PL quantum yields of the dimers and the monomers. It is often not possible to measure such very low quantum yields in solution using conventional techniques, and we show here that PTOFS provides access to this information. The quantum yield of the dimers was found to be of same order of magnitude as that of the monomers, of the order of 10−6, which indicates that the interparticle ‘electromagnetic hot-spots’ do not play a major role in the luminescence emission mechanism in such plasmonic molecules.

 Please wait while we load your content...
Please wait while we load your content...