Functionalization and characterization of electrocrystallized iron oxide nanoparticles in the presence of β-cyclodextrin
Abstract
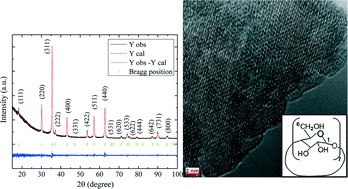
Electrocrystallized iron oxide nanoparticles were prepared by a chronoamperometric technique in the presence of β-cyclodextrin. The electrocrystallization process was performed with two iron electrodes in an electrolytic bath. The effect of β-cyclodextrin concentration, applied potential and bath temperature on the structural properties and magnetization of the nanoparticles was studied. FT-IR spectroscopy, X-ray diffraction, electron microscopy, magnetometry and Mössbauer spectroscopy were used to characterize the samples. XRD patterns confirmed the formation of the spinel Fe3O4 crystal structure. FT-IR spectra confirmed the presence of organic molecules at the surface of the particles. Electron microscopy images showed that the mean particle size is in the range 20–80 nm. Based on these images, we found that tuning the growth conditions has a strong effect on the particle size and morphology. High-resolution transmission electron microscopy images showed the aggregation of very fine crystallites with different orientations. The lattice striations confirmed the well-crystallized nature of the nanoparticles. The ring-like electron diffraction patterns are attributed to diffraction from the crystal planes of iron oxide nanoparticles. Room-temperature magnetization loops showed that all samples are magnetically soft with very little hysteresis, but the specific magnetization ranging from 14–80 A m2 kg−1 is highly dependent on the particle size and the experimental conditions. The room-temperature Mössbauer spectra are typical of non-stoichiometric Fe3−δO4, with a small excess of Fe3+ (0.07 ≤ δ ≤ 0.18). Our results showed that it is possible to improve the crystal structure of the particles by tuning the growth parameters.

 Please wait while we load your content...
Please wait while we load your content...