Efficient DBU accelerated synthesis of 18F-labelled trifluoroacetamides†
Abstract
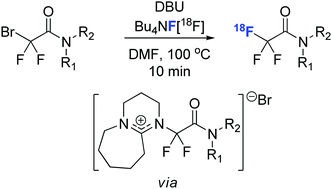
Nucleophilic 18F-fluorination of bromodifluoromethyl derivatives was performed using [18F]Bu4NF in the presence of DBU (1,8-diazabicyclo[5.4.0]undec-7-ene). This novel procedure provided a diverse set of [18F]trifluoroacetamides in good to excellent radiochemical conversions. A mechanism where DBU acts as organomediator in this transformation is proposed.


 Please wait while we load your content...
Please wait while we load your content...