Synthesis and characterization of a novel injectable alginate–collagen–hydroxyapatite hydrogel for bone tissue regeneration
Abstract
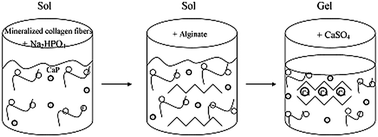
The rigid architecture of implanted scaffolds for bone tissue engineering often provides a limited ability to fill irregular contours of bone defects. Thus, injectable hydrogels are used to completely fill the defects while enhancing bone formation of the area. In this study, an injectable alginate hydrogel with a gelation time ranging from 5–10 minutes was developed by varying the concentrations of phosphate and calcium involved in the gelation process. The incorporation of mineralized collagen fibers within the hydrogel further increased the mechanical properties and osteoconductivity of the hydrogels. The gelation time, dynamic mechanical analysis (DMA) and thermogravimetric analysis (TGA) results suggested that the order in which the phosphate was added to the system had an effect on the gelation mechanism. This was further investigated to find that the addition of phosphate prior to the alginate powder resulted in better control of the gelation time and thus a more uniform hydrogel. The presence of hydroxyapatite in the hydrogels was confirmed using various characterization techniques, including X-ray diffraction (XRD) and Fourier transform infrared spectroscopy (FTIR). This novel fabrication process allowed for the development of an injectable hydrogel system with components necessary for promoting enhanced bone regeneration as well as host-implant integration.

 Please wait while we load your content...
Please wait while we load your content...