Heterostructured Cu2O/CuO decorated with nickel as a highly efficient photocathode for photoelectrochemical water reduction†
Abstract
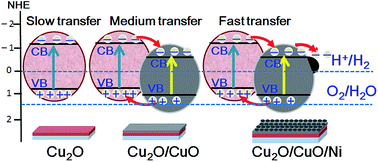
Here we report the design, synthesis and characterization of a novel Cu2O/CuO heterojunction decorated with a nickel cocatalyst as a highly efficient photocathode for solar hydrogen production. The heterojunction structure was shown and examined by X-ray absorption spectroscopy, X-ray photoelectron spectroscopy (XPS), Raman spectroscopy and Tip-enhanced Raman spectroscopy (TERS). Due to the synergistic effect, the Cu2O/CuO heterojunction gave a remarkably improved photocurrent density (−2.1 mA cm−2), i.e. 3.1 times higher than a Cu2O photoelectrode. Additionally, the Cu2O/CuO heterojunction, when decorated with the nickel cocatalyst, showed six-fold and two-fold increases in photocurrent density (−4.3 mA cm−2) respectively when compared to Cu2O and the bare Cu2O/CuO at 0 V vs. RHE under AM 1.5 illumination (100 mW cm−2). Interestingly, the Ni decorated Cu2O/CuO photocathode showed an impressive solar conversion efficiency of 2.71% at −0.4 V vs. Pt, i.e. 467% higher compared to the bare Cu2O/CuO. After 20 minutes of standard solar illumination, 87.7% of the initial photocurrent density was retained for the nickel decorated Cu2O/CuO heterojunction. This is more than 1.5 times that of the bare Cu2O/CuO (53.6%), suggesting that surface modification with Ni not only effectively promotes water splitting but also stabilizes the photoelectrode. The enhanced photoelectrochemical performance is attributable to the efficient charge transfer and protective role of Ni, the improved crystallinity and the synergistic effect of the heterojunction in light absorption and charge separation. This inexpensive photocathode with increased photocurrent density and photostability offers a higher promise for solar hydrogen production.


 Please wait while we load your content...
Please wait while we load your content...