Sulfur double locked by a macro-structural cathode and a solid polymer electrolyte for lithium–sulfur batteries
Abstract
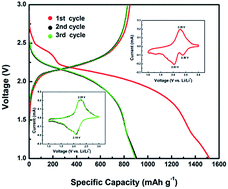
Both macro-structural cathode materials and a metal–organic framework (MIL-53(Al)) modified solid polymer electrolyte are used to inhibit polysulfide dissolution and shuttling in all-solid-state lithium–sulfur batteries. At 80 °C, the discharge capacities of 1520 mA h g−1 in the first cycle at 0.2 C and 325 mA h g−1 in the 1000th cycle at 4 C are obtained, demonstrating exceptional high rate capability and long-term cycling performance of the batteries. Mechanisms for such an enhancement are investigated by cyclic voltammetry, X-ray photoelectron spectroscopy (XPS), electrochemical impedance spectroscopy (EIS), and thermo-gravimetric analysis techniques. In the EIS spectra there is no semicircle caused by the polysulfide shuttling and reaction at the electrolyte/Li interface, proving that polysulfide dissolution is adequately inhibited. The XPS spectra on the Li anode surface after 10 discharge/charge cycles show that sulfur and polysulfide do not transfer from the cathode to the anode. The –C–S– bond in the original cathode is observed by XPS, indicating that sulfur is linked to macro-structural cathode materials by the thermal treatment. In addition, an intermediate forms during cycling and displays steady electrochemical reversibility. These data indicate that the macro-structural cathode and solid polymer electrolyte play crucial roles in blocking polysulfide dissolution and shuttling, and lead to the outstanding cycling performance of all-solid-state lithium–sulfur batteries.

 Please wait while we load your content...
Please wait while we load your content...