Tandem redox mediator/Ni(ii) trihalide complex photocycle for hydrogen evolution from HCl†
Abstract
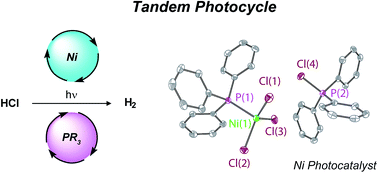
Photoactivation of M–X bonds is a challenge for photochemical HX splitting, particularly with first-row transition metal complexes because of short intrinsic excited state lifetimes. Herein, we report a tandem H2 photocycle based on combination of a non-basic photoredox phosphine mediator and nickel metal catalyst. Synthetic studies and time-resolved photochemical studies have revealed that phosphines serve as photochemical H-atom donors to Ni(II) trihalide complexes to deliver a Ni(I) centre. The H2 evolution catalytic cycle is closed by sequential disproportionation of Ni(I) to afford Ni(0) and Ni(II) and protolytic H2 evolution from the Ni(0) intermediate. The results of these investigations suggest that H2 photogeneration proceeds by two sequential catalytic cycles: a photoredox cycle catalyzed by phosphines and an H2-evolution cycle catalyzed by Ni complexes to circumvent challenges of photochemistry with first-row transition metal complexes.

 Please wait while we load your content...
Please wait while we load your content...