Durable hydrogen evolution from water driven by sunlight using (Ag,Cu)GaSe2 photocathodes modified with CdS and CuGa3Se5†
Abstract
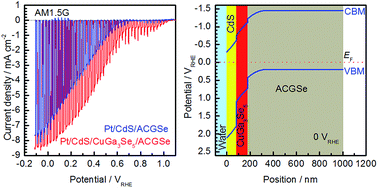
The photoelectrochemical properties of (Ag,Cu)GaSe2 (ACGSe) modified by deposition of CdS and CuGa3Se5 layers were investigated. The CdS and CuGa3Se5 layers formed p–n junctions with an appropriate band diagram on the surface of the electrode and they clearly increased the cathodic photocurrent and onset potential. The Pt, CdS, and CuGa3Se5 modified ACGSe (Pt/CdS/CuGa3Se5/ACGSe) with an appropriate thickness of CuGa3Se5 layers (ca. 100 nm) showed a cathodic photocurrent of 8.79 mA cm−2 at 0 VRHE and an onset potential of 0.62 VRHE (defined as cathodic photocurrent of 1.0 mA cm−2) under simulated sunlight irradiation in 0.1 M Na2HPO4 (pH 10). Pt/CdS/CuGa3Se5/ACGSe showed durable cathodic current under the observed reaction conditions and hydrogen was evolved for about 20 days.
- This article is part of the themed collections: Global Energy Challenges: Hydrogen Energy and Global Energy Challenges: Energy Applications

 Please wait while we load your content...
Please wait while we load your content...