Diazo compounds for the bioreversible esterification of proteins†
Abstract
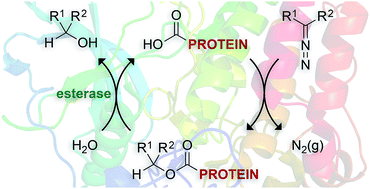
A diazo compound is shown to convert carboxylic acids to esters efficiently in an aqueous environment. The basicity of the diazo compound is critical: low basicity does not lead to a reaction but high basicity leads to hydrolysis. This reactivity extends to carboxylic acid groups in a protein. The ensuing esters are hydrolyzed by human cellular esterases to regenerate protein carboxyl groups. This new mode of chemical modification could enable the key advantages of prodrugs to be translated from small-molecules to proteins.

 Please wait while we load your content...
Please wait while we load your content...