Modification of Rapana thomasiana hemocyanin with choline amino acid salts significantly enhances its antiproliferative activity against MCF-7 human breast cancer cells†
Abstract
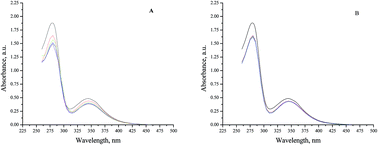
This is the first study on the interactions of ionic liquids with large metalloproteins, in particular hemocyanins (Hcs). At first, complexes of a Hc from Rapana thomasiana (RtH) with a series of biocompatible choline amino acid salts [Chol][AA] were obtained. Applying UV-vis spectroscopy, Fourier-transformed infrared spectroscopy and differential scanning calorimetry the effect of these organic salts on the structure and thermal stability of RtH was assessed. Then, the cytotoxic effect of RtH–[Chol][AA] on breast cancer cells (MCF-7) and 3T3 fibroblast cells (non cancerous) was evaluated. We found that all [Chol][AA] induced clear time- and concentration-dependent alterations in the RtH conformation. The conformation and the thermal stability of IL-modified RtH depend strongly on the type of the anion of the tested compounds. All [Chol][AA]-modified RtHs exhibited lower thermal stability than the native RtH. At the same time, we established a good correlation between the structure of RtH and its antitumor activity. Namely, RtH–[Chol][AA] complexes exhibited enhanced antiproliferative activity toward the MCF-7 cell line. The observed antiproliferative effect was cell specific and the compounds have no effect or in some cases have stimulatory effect on fibroblasts.

 Please wait while we load your content...
Please wait while we load your content...