Synthesis of g-C3N4/Bi2O3/TiO2 composite nanotubes: enhanced activity under visible light irradiation and improved photoelectrochemical activity†
Abstract
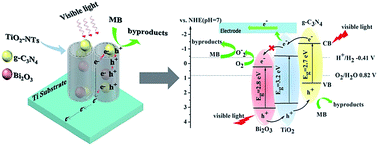
g-C3N4 and Bi2O3 were successfully incorporated into TiO2 nanotubes (TiO2-NT) to produce a composite nanotube material designated as g-C3N4/Bi2O3/TiO2. The photoelectrochemical (PEC) activity under visible light irradiation with respect to bleaching of methylene blue (MB) and degradation of phenol were determined. The UV-vis absorption spectrum of the composite material, g-C3N4/Bi2O3/TiO2-NT, was red-shifted toward a narrower band gap energy (Eg). The valence band (VB) of g-C3N4/Bi2O3/TiO2-NT was shifted to a more positive potential resulting in an increased driving force for water oxidation. The photocurrent generated by g-C3N4/Bi2O3/TiO2-NTs was approximately 15 times higher and the incident photon-to-current efficiency (IPCE at λ = 400 nm) was higher than that of the naked TiO2-NTs. This response is attributed to increasing the lifetimes of photo-generated electron–hole pairs. A significantly higher PEC response in terms of the bleaching of MB was observed on g-C3N4/Bi2O3/TiO2-NT. The roles of improved charge separation and subsequent higher electron-transfer efficiencies for in g-C3N4/Bi2O3/TiO2-NT electrodes were examined.

 Please wait while we load your content...
Please wait while we load your content...