Evaluating chemical ligation techniques for the synthesis of block copolypeptides, polypeptoids and block copolypept(o)ides: a comparative study†
Abstract
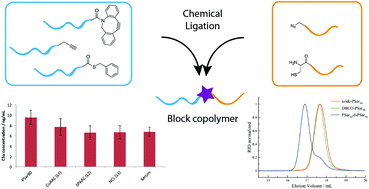
In this work, we describe the synthesis of block copolypeptides, polypeptoids and block copolypept(o)ides by chemical ligation techniques. Polysarcosine (PSar), poly(N-ε-trifluoroacetyl-L-lysine) (PLys(TFA)) and poly(γ-benzyl-L-glutamate) (PGlu(OBzl)) homopolymers of different polarities and end group functionalities but with similar average degrees of polymerization (Xn = 50 and 100) could be obtained by ring opening polymerization (ROP) of α-amino acid N-carboxyanhydrides (NCA) and postpolymerization modification reactions. In the next step, these polymers were applied to copper(I)-catalyzed azide–alkyne coupling (CuAAC), strain-promoted azide–alkyne coupling (SPAAC) and native chemical ligation (NCL). Our results suggest that all the employed ligation techniques can be used for the synthesis of block copolypeptides, polypeptoids and block copolypept(o)ides. SPAAC displayed, for most conditions, the highest ligation efficiencies (up to 86%) and, from a practical point of view, is the most feasible method. NCL, however, performed very well for short hydrophilic polymers (up to 88%) and is favourable for the ligation of peptides from solid phase peptide synthesis (SPPS) to polysarcosine. As a proof of principle, we report a protocol for an efficient NCL coupling of polysarcosine to the T-cell receptor core peptide (TCR CP), which is known to inhibit IL-2 production in antigen-stimulated T cells and, therefore, to suppress inflammation. In a comparative study, the ligation method, which directly relates to the chemical nature of the ligation site, neither influenced cytotoxicity nor complement activation. To conclude, chemical ligation techniques represent a complementary synthetic approach to the well-established sequential ring opening polymerization of NCAs.

 Please wait while we load your content...
Please wait while we load your content...