A passive-flow microfluidic device for imaging latent HIV activation dynamics in single T cells†
Abstract
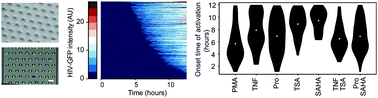
Quantifying cell-to-cell variability in drug response dynamics is important when evaluating therapeutic efficacy. For example, optimizing latency reversing agents (LRAs) for use in a clinical “activate-and-kill” strategy to purge the latent HIV reservoir in patients requires minimizing heterogeneous viral activation dynamics. To evaluate how heterogeneity in latent HIV activation varies across a range of LRAs, we tracked drug-induced response dynamics in single cells via live-cell imaging using a latent HIV–GFP reporter virus in a clonal Jurkat T cell line. To enable these studies in suspension cells, we designed a simple method to capture an array of single Jurkat T cells using a passive-flow microfluidic device. Our device, which does not require external pumps or tubing, can trap hundreds of cells within minutes with a high retention rate over 12 hours of imaging. Using this device, we quantified heterogeneity in viral activation stimulated by transcription factor (TF) activators and histone deacetylase (HDAC) inhibitors. Generally, TF activators resulted in both faster onset of viral activation and faster rates of production, while HDAC inhibitors resulted in more uniform onset times, but more heterogeneous rates of production. Finally, we demonstrated that while onset time of viral gene expression and rate of viral production together predict total HIV activation, rate and onset time were not correlated within the same individual cell, suggesting that these features are regulated independently. Overall, our results reveal drug-specific patterns of noisy HIV activation dynamics not previously identified in static single-cell assays, which may require consideration for the most effective activate-and-kill regime.

 Please wait while we load your content...
Please wait while we load your content...