Quantitative structure–activity relationship correlation between molecular structure and the Rayleigh enantiomeric enrichment factor†
Abstract
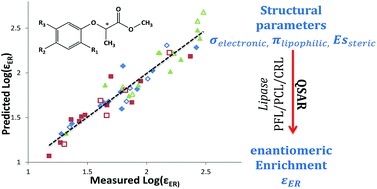
It was recently demonstrated that under environmentally relevant conditions the Rayleigh equation is valid to describe the enantiomeric enrichment – conversion relationship, yielding a proportional constant called the enantiomeric enrichment factor, εER. In the present study we demonstrate a quantitative structure–activity relationship model (QSAR) that describes well the dependence of εER on molecular structure. The enantiomeric enrichment factor can be predicted by the linear Hansch model, which correlates biological activity with physicochemical properties. Enantioselective hydrolysis of sixteen derivatives of 2-(phenoxy)propionate (PPMs) have been analyzed during enzymatic degradation by lipases from Pseudomonas fluorescens (PFL), Pseudomonas cepacia (PCL), and Candida rugosa (CRL). In all cases the QSAR relationships were significant with R2 values of 0.90–0.93, and showed high predictive abilities with internal and external validations providing QLOO2 values of 0.85–0.87 and QExt2 values of 0.8–0.91. Moreover, it is demonstrated that this model enables differentiation between enzymes with different binding site shapes. The enantioselectivity of PFL and PCL was dictated by electronic properties, whereas the enantioselectivity of CRL was determined by lipophilicity and steric factors. The predictive ability of the QSAR model demonstrated in the present study may serve as a helpful tool in environmental studies, assisting in source tracking of unstudied chiral compounds belonging to a well-studied homologous series.

 Please wait while we load your content...
Please wait while we load your content...