The synergistic effect in Co–Ce oxides for catalytic oxidation of diesel soot†
Abstract
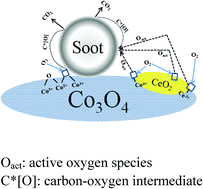
Diesel soot, a major contributor to PM emission, severely threatens human health and the environment. Catalytic oxidation of trapped diesel soot is a feasible solution to alleviating this problem. In this study, highly active Co–Ce catalysts were synthesized by the citric acid complex method and their catalytic behaviour was investigated by means of TG analysis. The best catalytic performance was acquired over the Co0.93Ce0.07 catalyst with T10 = 315 °C and T50 = 370 °C. These catalyst samples were also characterized by XRD, Raman spectrum, soot-TPR, H2-TPR, O2-TPD, XPS and in situ Raman spectra. Cerium cations were partially incorporated into the lattice of Co3O4 in Co–Ce mixed oxides, which led to the lattice expansion. This structure modification brought about a series of changes in the surface and the lattice of these catalysts. The capability of oxygen adsorption and desorption was slightly improved, and the redox ability of mixed oxides was intensified owing to the strong interaction between Co3O4 and CeO2. Surface lattice oxygen and bulk lattice oxygen could also be activated at a lower temperature. Moreover, active oxygen species (superoxide and peroxide) as well as carbon–oxygen intermediates (carbonyl and formate species) were discovered in soot combustion. Reactions for producing formate species were important steps for the soot–O2 reaction. The enhanced effects of both redox mechanism and oxygen spillover mechanism were probably due to the synergistic effect between the two oxides.

 Please wait while we load your content...
Please wait while we load your content...