Adsorption of CO2 on amine-functionalised MCM-41: experimental and theoretical studies†
Abstract
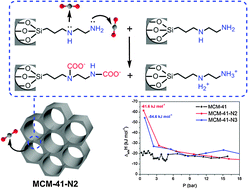
Adsorption of CO2 on MCM-41 functionalised with [3-(2-aminoethylamino)propyl]trimethoxysilane (MCM-41-N2), N1-(3-trimethoxysilylpropyl)diethylenetriamine (MCM-41-N3), 4-aminopyridine (MCM-41-aminopyridine), 4-(methylamino)pyridine (MCM-41-methylaminopyridine) and 1,5,7-triazabicyclo[4.4.0]dec-5-ene (MCM-41-guanidine) was investigated. The amine-functionalised materials were characterised by 29Si and 13C solid-state nuclear magnetic resonance, N2 adsorption/desorption isotherms, X-ray diffraction and transmission electron microscopy. CO2 adsorption at 1.0 bar and 30 °C showed that the amount of CO2 (nads/mmol g−1) adsorbed on MCM-41-N2 and MCM-41-N3 is approximately twice the amount adsorbed on MCM-41. For MCM-41-aminopyridine, MCM-41-methylaminopyridine and MCM-41-guanidine, the CO2 adsorption capacity was smaller than that of MCM-41 at the same conditions. The proton affinity (computed with wB97x-D/6-311++G(d,p)) of the secondary amino groups is higher than that of the primary amino groups; however, the relative stabilities of the primary and secondary carbamates are similar. The differential heat of adsorption decreases as the number of secondary amino groups increases.

 Please wait while we load your content...
Please wait while we load your content...