Ion pair based dispersive liquid–liquid microextraction for the preconcentration of ultra-trace levels of bismuth(iii) and its determination by electrothermal atomic absorption spectroscopy
Abstract
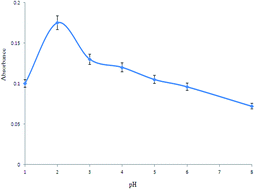
A simple, selective and efficient ion pair based dispersive liquid–liquid microextraction method was used for the preconcentration of ultra-trace levels of Bi(III) prior to its determination by electrothermal atomic absorption spectrometry. This method is based on the complexation of Bi(III) with thiocyanate anion to form the anionic complexes, Bi(SCN)63− and Bi(SCN)4−. A cationic surfactant, cetylpyridinium chloride (CPC), was used as a counter ion. The resulting ion pair complexes were extracted into fine droplets of carbon tetrachloride by rapid injection of a mixture of acetone (as the disperser solvent) and carbon tetrachloride (as the extraction solvent) into the sample solution. Important parameters affecting the extraction efficiency, including sample pH, thiocyanate and CPC concentrations, and type and volume of the extraction and disperser solvents, were examined and optimized. Under the optimum conditions, the calibration graph was linear in the range of 0.3–8 μg L−1 Bi (III) with a correlation coefficient of 0.9979. The relative standard deviation (RSD, %) based on eight replicate analyses of 2 μg L−1 Bi(III) was 4.8%, and the detection limit (DL) of Bi(III) was 0.07 μg L−1. The proposed method was validated by the analysis of a certified reference material and the spike method. The obtained results were in very good agreement with the certified values. The proposed method was successfully applied for the determination of ultra-trace levels of Bi(III) in different water and human serum samples.

 Please wait while we load your content...
Please wait while we load your content...