Hydrothermal synthesis of nitrogen-doped graphene hydrogels using amino acids with different acidities as doping agents†
Abstract
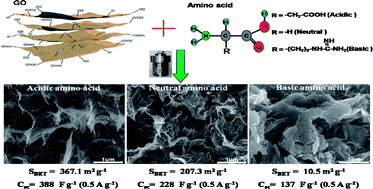
A one-pot hydrothermal route was developed for the preparation of nitrogen-doped graphene (NG) hydrogels using graphene oxide (GO) as a raw material and nine amino acids with different acidities (acidic, neutral and basic) as doping agents. The morphology, structure and composition of the prepared NG using three amino acids (DL-aspartic acid, acidic; L-glycine, neutral; L-arginine, basic) were characterized by SEM, nitrogen physisorption, Raman and XPS spectroscopy. Acidic amino acids yielded NG with a cross-linked 3D network with a large specific surface area of 367.1 m2 g−1, while NG arising from the use of basic amino acids showed a tightly stacked structure with a much smaller surface area of 10.5 m2 g−1. The charged amino acids, and the ensuing electrostatic interactions between the amino acid and GO, affect the morphology of NG, and ultimately affect its electrochemical performance. The samples prepared using acidic amino acids, with the lowest surface N content (1.0%) but the largest surface area, displayed high specific capacitance of 246 F g−1 at 3 A g−1. The microstructure, surface area and effective nitrogen content, mainly the pyridinic nitrogen group related to pseudocapacitance, play important roles in the capacitive performance of the NG samples.

 Please wait while we load your content...
Please wait while we load your content...