Reversible CO2 binding triggered by metal–ligand cooperation in a rhenium(i) PNP pincer-type complex and the reaction with dihydrogen†
Abstract
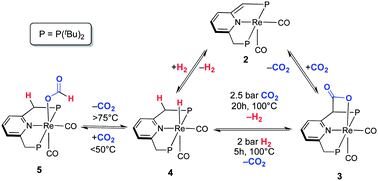
Herein we report the reversible binding of CO2 to a very rare dearomatized PNP pincer complex (cis-[Re(PNPtBu*)(CO)2] 2). The [1,3]-addition of CO2 to the pincer complex is triggered by metal–ligand cooperation via an aromatization/dearomatization sequence to form cis-[Re(PNPtBu–COO)(CO)2] (3) via Re–O and C–C bond formation. The reversible binding was demonstrated by the exchange reaction of 13CO2/CO2 from the isotopically labelled compound cis-[Re(PNPtBu–13COO)(CO)2] (3a). Furthermore, complex 3 reacts with H2 to give free CO2 and the aromatized hydride complex cis-[Re(PNPtBu)(CO)2H] (4), which undergoes the reverse reaction to re-form 3 under CO2 pressure at elevated temperature. Alternatively, 4 reacts, in a low temperature pathway, via the insertion of CO2 into the Re–H bond to give the formate complex cis-[Re(PNPtBu)(CO)2(OOCH)] (5)]. Remarkably, complex 5 catalyses efficiently the dehydrogenation of formic acid under base-free conditions.

 Please wait while we load your content...
Please wait while we load your content...