β-Substituted γ-butyrolactams from mucochloric acid: synthesis of (±)-baclofen and other γ-aminobutyric acids and useful building blocks†
Abstract
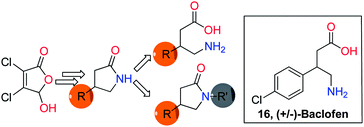
In the course of our exploration of the application of mucohalic acids in organic synthesis, we reported the synthesis of α,β-dihalo-α,β-unsaturated γ-butyrolactams by reductive amination of a suitable mucohalic acid and a suitable amine. However, the functionalization of the α- and/or β-halogen was found to be elusive under Suzuki conditions that worked well with the corresponding γ-lactones. Although the corresponding β-aryl derivative was obtained under some of the modified Suzuki conditions tried, the yields were found to be very low. Next, the Suzuki coupling was performed on mucochloric acid prior to the reductive amination step. By careful control of the reaction conditions, only β-substitution was achieved to yield the corresponding β-aryl mucochloric acid. This could then be converted to the corresponding β-substituted γ-butyrolactam by reductive amination with 2,4-dimethoxybenzylamine (DMB-NH2) followed by reduction by nickel boride and subsequent deprotection of the DMB group. The resulting γ-butyrolactam could either be alkylated or hydrolyzed resulting in the corresponding β-substituted γ-aminobutyric acid. This methodology was extended to synthesize (±)-baclofen in five steps starting from mucochloric acid.

 Please wait while we load your content...
Please wait while we load your content...