NHC-catalyzed homoenolate reaction of enals and nitroalkenes: computational study of mechanism, chemoselectivity and stereoselectivity†
Abstract
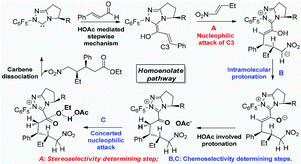
A systematic theoretical study has been carried out on the NHC-catalyzed homoenolate reaction of enals and nitroalkenes. The detailed mechanism of these nitroalkenes participated homoenolate reactions, the chemoselectivity of the homoenolate (vs. Stetter) pathway and the intriguing syn- (vs. anti-) stereoselectivity have been investigated. Calculations show that the homoenolate and Stetter reaction (Path-homo and Path-set) first undergo the same formation process of the Breslow intermediate, which proceeds via a HOAc mediated stepwise mechanism. The subsequent process of Path-homo consists of five steps: nucleophilic attack of the Breslow intermediate (C3 atom) on the nitroalkene, intramolecular proton transfer, HOAc involved protonation of C2, concerted nucleophilic attack of OEt on the carbonyl, and final carbene dissociation. The subsequent process of Path-set consists of three steps: nucleophilic attack of the Breslow intermediate (C1 atom) on the nitroalkene, intramolecular proton transfer and carbene dissociation. Path-homo is more feasible than Path-set. The highly exoergonic nucleophilic attack, the facile intramolecular proton transfer and attack of OEt on the carbonyl in Path-homo result in its feasibility. The stereoselectivity determining step of Path-homo is the attack of the Breslow intermediate on the nitroalkene, in which the steric hindrance and hydrogen bonding determine the syn stereoselectivity.

 Please wait while we load your content...
Please wait while we load your content...