Mercaptobenzoic acid-palladium(0) complexes as active catalysts for S-benzylation with benzylic alcohols via (η3-benzyl)palladium(ii) cations in water†
Abstract
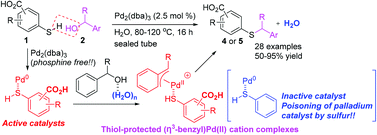
Mercaptobenzoic acid-palladium(0) complexes show high catalytic activity for S-benzylation with benzylic alcohols via the (η3-benzyl)palladium(II) cation in water. Notably, these palladium(0) complexes could play an important role in formation of active (η3-benzyl)palladium(II) cation complexes followed by S-benzylation. Hammett studies on the rate constants of S-benzylation by various substituted alcohols show good correlation between log(kX/kH) and the σ+ value of the respective substituents. From the slope, negative ρ values are obtained, suggesting that there is a build-up of positive charge in the transition state. Water plays an important role in the catalytic system for sp3 C–O bond activation and stabilization of the activated Pd(II) cation species. The catalytic system can be performed using only 2.5 mol% Pd2(dba)3 without the phosphine ligand or other additives.

 Please wait while we load your content...
Please wait while we load your content...