Cerium-based M4L4 tetrahedrons containing hydrogen bond groups as functional molecular flasks for selective reaction prompting†
Abstract
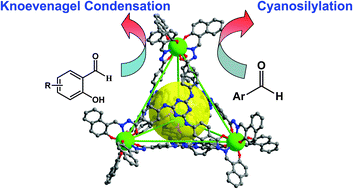
The application of metal–organic polyhedrons as “molecular flasks” has resulted in a surge of interest in the reactivity and properties of molecules within their well-defined cavity. Inspired by the structures of the natural enzymatic pockets, two neutral metal–organic tetrahedrons, Ce-TBMN and Ce-TBAS, were achieved via self-assembly by incorporating triamine-triazine and amide groups as hydrogen bond sites into the fragments of the ligands respectively. Both of them could act as molecular flasks to prompt the Knoevenagel condensation reactions of salicylaldehyde derivatives and cyanosilylation reactions of aromatic aldehydes. Experiments of catalysts with different cavity radii and substrates with different sizes and shapes, as well as competitive experiments using nonreactive guests as inhibitors demonstrated that the tetrahedrons exhibited enzymatically catalytic behavior and the catalytic reactions occurred in the “molecular flasks”. Control experiments with the H6TMBN or H6TBAS ligands themselves as the catalyst in the Knoevenagel condensation were carried out under the same conditions. For the smaller substrates, their conversions catalyzed by the ligands were significantly lower than those catalyzed by Ce-TBMN or Ce-TBAS, respectively, suggesting that metal–organic polyhedrons could effectively fix multi hydrogen bond groups to avoid the “self-quenching” effect, enhancing the catalytic activity of the multi-hydrogen bond groups in the homogeneous state.

 Please wait while we load your content...
Please wait while we load your content...