Borohydrides: from sheet to framework topologies†
Abstract
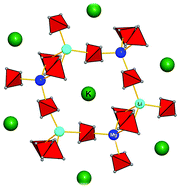
The five novel compounds ALiM(BH4)4 (A = K or Rb; M = Mg or Mn) and K3Li2Mg2(BH4)9 crystallizing in the space groups Aba2 and P2/c, respectively, represent the first two-dimensional topologies amongst homoleptic borohydrides. The crystal structures have been solved, refined and characterized by synchrotron X-ray powder diffraction, neutron powder diffraction and solid-state DFT calculations. Minimal energies of ordered models corroborate crystal symmetries retrieved from diffraction data. The layered Li–Mg substructure forms negatively charged uninodal 4-connected networks. It is shown that this connectivity cannot generate the long sought-after, bimetallic Li–Mg borohydrides without countercations when assuming preferred coordination polyhedra as found in Mg(BH4)2 and LiBH4. The general properties of the trimetallic compound series are analogous with the anhydrous aluminosilicates. Additionally, a relationship with zeolites is suggested, which are built from three-dimensional Al–Si–O networks with a negative charge on them. The ternary metal borohydride systems are of interest due to their potential as novel hydridic frameworks and will allow exploration of the structural chemistry of light-metal systems otherwise subject to eutectic melting.

 Please wait while we load your content...
Please wait while we load your content...