An atom in molecules study of infrared intensity enhancements in fundamental donor stretching bands in hydrogen bond formation†
Abstract
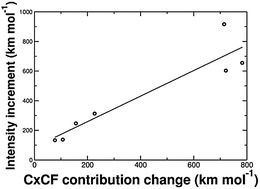
Vibrational modes ascribed to the stretching of X–H bonds from donor monomers (HXdonor) in complexes presenting hydrogen bonds (HF⋯HF, HCl⋯HCl, HCN⋯HCN, HNC⋯HNC, HCN⋯HF, HF⋯HCl and H2O⋯HF) exhibit large (4 to 7 times) infrared intensity increments during complexation according to CCSD/cc-pVQZ-mod calculations. These intensity increases are explained by the charge–charge flux–dipole flux (CCFDF) model based on multipoles from the Quantum Theory of Atoms in Molecules (QTAIM) as resulting from a reinforcing interaction between two contributions to the dipole moment derivatives with respect to the vibrational displacements: charge and charge flux. As such, variations that occur in their intensity cross terms in hydrogen bond formation correlate nicely with the intensity enhancements. These stretching modes of HXdonor bonds can be approximately modeled by sole displacement of the positively charged hydrogens towards the acceptor terminal atom with concomitant electronic charge transfers in the opposite direction that are larger than those occurring for the H atom displacements of their isolated donor molecules. This analysis indicates that the charge–charge flux interaction reinforcement on H-bond complexation is associated with variations of atomic charge fluxes in both parent molecules and small electronic charge transfers between them. The QTAIM/CCFDF model also indicates that atomic dipole flux contributions do not play a significant role in these intensity enhancements.

 Please wait while we load your content...
Please wait while we load your content...