Synthesis and structural characterization of three 3-D aluminogermanates with different topologies†
Abstract
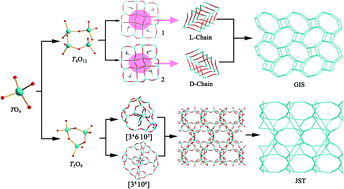
Three 3-D aluminogermanates L-[C2NH8][AlGe3O8] (1), D-[C2NH8][AlGe3O8] (2) and [Ni(en)3][Al3Ge3O12(H2en)0.5] (3) (en = 1,2-ethanediamine) have been successfully synthesized through a hydrothermal synthesis method (1 and 2 were obtained as a mixture in one autoclave). Crystal structural analysis reveals that three compounds, 1, 2 and 3, are made up of GeO4 (AlO4) tetrahedra. Compounds 1 and 2 with a typical GIS topology are composed of 4-rings, while 3 is constructed exclusively from 3-rings with a JST topology. Compounds 1 and 2 are enantiomers and crystallize in chiral space groups P43212 and P41212, respectively. In compound 3, rigid chiral transition-metal complex cations, [Ni(en)3]2+, and protonated ethylenediamine cations work together as structure directing agents (SDAs) and induce two different kinds of cages ([38·106] and [34·6·103]), which further construct the final structure.

 Please wait while we load your content...
Please wait while we load your content...